The USZ is the first hospital in Switzerland to offer a new blood test based on genetic biomarkers that significantly improves the early detection of aggressive prostate cancer. Several scientific studies with a total of over 75,000 participants prove the high accuracy of the test, which has been used in Sweden and Norway since 2017.
Prostate cancer is the most common cancer in men in Switzerland. The number of cases is also steadily increasing due to the ageing population. Early detection, for example as part of regular cancer screening, is important. It increases the chances of successfully treating aggressive tumors and, ideally, curing the cancer.
PSA and DRU: the disadvantages of the current diagnostic routine
For years, the PSA value and the digital rectal examination (DRU) have formed the basis of prostate cancer screening in everyday urological practice. Both methods are the subject of intense scientific debate. The PSA value is not a specific cancer marker and can be influenced by a variety of factors (sexual intercourse, cycling, inflammation, benign enlargement of the prostate, certain medications, TUR-P). The PSA value is also elevated in prostate carcinoma.
Stockholm3 test: New biomarkers enable greater precision
As early as 2022, a political recommendation was made in the EU to implement new promising tests in screening programs. These tests include the Stockholm3 test, which was developed at the renowned Karolinska Institute in Sweden. It has been used in Scandinavia for the early detection of prostate cancer since 2017. It is now also recommended in the Swedish guidelines for prostate cancer.
The Stockholm3 test significantly improves the precision of the diagnosis compared to PSA screening and also compared to the Rotterdam Prostate Cancer Risk Calculator (RPCRC): the new test finds 89% (Viste et al., 2020) more cases of aggressive prostate cancer, while at the same time reducing unnecessary prostate biopsies by 76% (Eklund et al., 2019). The success of the innovative test has been demonstrated in numerous studies with a total of over 75,000 participants. Five international awards from leading oncology journals and urological societies underline the medical benefits of the new test procedure.
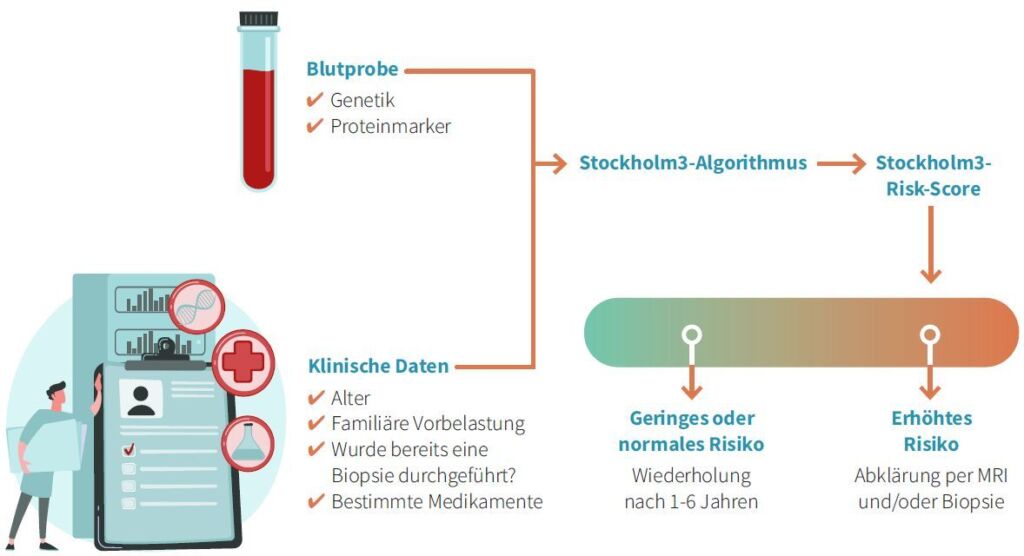
How the new test works
The innovative blood test calculates an STHLM3 value from a combination of plasma proteins, genetic polymorphisms and clinical variables to assess the risk of prostate cancer.
With the value of the risk assessment, the doctor also receives a recommendation as to whether further investigations such as prostate biopsy and/or MRI are necessary. In addition to diagnosing prostate cancer, the test can also indicate whether the cancer is aggressive and active treatment should be started as soon as possible. Further genetic stratification of the removed prostate tissue with other tests available at the USZ can further personalize the treatment options.
As a referring physician, how can you use the new prostate cancer screening at the USZ?
The new test is recommended for men between the ages of 45 and 75. It is only used for early detection and initial diagnosis, but not if prostate cancer has already been detected. Patients undergoing cancer screening or with suspected prostate cancer can be referred to the Department of Urology. After a consultation with our urologists, we order the blood test. For this purpose, blood is taken from the patient once and sent to the relevant laboratory for analysis. The result is usually available to the USZ within 3 weeks of dispatch and is discussed with the patient at the USZ.
Do you still have questions?
You are welcome to contact us if you have any questions about the test and the diagnosis of your patients.
